Multiple Choice
Identify the
choice that best completes the statement or answers the question.
|
|
|
1.
|
For this pair of items, choose the option that best
describes their relationship.
(A) The number of alpha glucose 1-4 linkages in cellulose
(B) The
number of alpha glucose 1-4 linkages in starch
a. | Item (A) is greater than item
(B). | b. | Item (A) is less than item
(B). | c. | Item (A) is exactly or very approximately equal
to item (B). | d. | Item (A) may stand
in more than one of the above relations to item (B). |
|
|
|
2.
|
For this pair of items, choose the option that best
describes their relationship.
(A) The probability of finding chitin in fungal cell walls
(B)
The probability of finding chitin in arthropod exoskeletons
a. | Item (A) is greater than item
(B). | b. | Item (A) is less than item
(B). | c. | Item (A) is exactly or very approximately equal
to item (B). | d. | Item (A) may stand
in more than one of the above relations to item (B). |
|
|
|
3.
|
For this pair of items, choose the option that best
describes their relationship.
(A) The number of cis double bonds in saturated fatty acids
(B)
The number of cis double bonds in unsaturated fatty acids
a. | Item (A) is greater than item
(B). | b. | Item (A) is less than item
(B). | c. | Item (A) is exactly or very approximately equal
to item (B). | d. | Item (A) may stand
in more than one of the above relations to item (B). |
|
|
|
4.
|
For this pair of items, choose the option that best
describes their relationship.
(A) The probability that amino acids with nonpolar side chains are
hydrophobic.
(B) The probability that amino acids with side chains containing a carboxyl group are
hydrophobic.
a. | Item (A) is greater than item
(B). | b. | Item (A) is less than item
(B). | c. | Item (A) is exactly or very approximately equal
to item (B). | d. | Item (A) may stand
in more than one of the above relations to item (B). |
|
|
|
5.
|
For this pair of items, choose the option that best
describes their relationship.
(A) The number of purines in the DNA strand
5'-AAGAGGAGAAA-3'
(B) The number of pyrimidines in the DNA strand
5'-AAGAGGAGAAA-3'
a. | Item (A) is greater than item
(B). | b. | Item (A) is less than item
(B). | c. | Item (A) is exactly or very approximately equal
to item (B). | d. | Item (A) may stand
in more than one of the above relations to item (B). |
|
|
|
6.
|
Which of the following is not a
polymer?
a. | glucose | b. | starch | c. | cellulose | d. | chitin | e. | DNA |
|
|
|
7.
|
What is the chemical mechanism by which cells make
polymers from monomers?
a. | phosphodiester linkages | b. | hydrolysis | c. | dehydration
reactions | d. | ionic bonding of monomers | e. | the formation of disulfide bridges between
monomers |
|
|
|
8.
|
How many molecules of water are needed to
completely hydrolyze a polymer that is 11 monomers long?
|
|
|
9.
|
Which of the following best summarizes the
relationship between dehydration reactions and hydrolysis?
a. | Dehydration reactions assemble polymers, and hydrolysis
reactions break down polymers. | b. | Macromolecular
synthesis occurs through the removal of water and digestion occurs through the addition of
water. | c. | Dehydration reactions can occur only after
hydrolysis. | d. | Hydrolysis creates
monomers, and dehydration reactions break down polymers. | e. | A and B are correct. |
|
|
|
10.
|
Which of the following polymers contain
nitrogen?
a. | starch | b. | glycogen | c. | cellulose | d. | chitin | e. | amylopectin |
|
|
|
11.
|
The molecular formula for glucose is 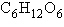 . What would be the molecular formula for a molecule made by linking three glucose
molecules together by dehydration reactions? . What would be the molecular formula for a molecule made by linking three glucose
molecules together by dehydration reactions?
|
|
|
12.
|
The enzyme amylase can break glycosidic linkages
between glucose monomers only if the monomers are the á form. Which of the following could
amylase break down?
a. | glycogen | b. | cellulose | c. | chitin | d. | A and B
only | e. | A, B, and C |
|
|
|
13.
|
On food packages, to what does the term
"insoluble fiber" refer?
a. | cellulose | b. | polypeptides | c. | starch | d. | amylopectin | e. | chitin |
|
|
|
14.
|
A molecule with the chemical formula
C6H12O6 is probably a
a. | carbohydrate. | b. | lipid. | c. | monosaccharide | d. | carbohydrate and
lipid only. | e. | carbohydrate and
monosaccharide only. |
|
|
|
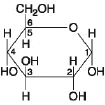
Figure
5.1
|
|
|
15.
|
If 2 molecules of the general type shown in Figure
5.1 were linked together, carbon 1 of one molecule to carbon 4 of the other, the single molecule that
would result would be
a. | maltose. | b. | fructose. | c. | glucose. | d. | galactose. | e. | sucrose. |
|
|
|
16.
|
Which of the following descriptors is true of the
molecule shown in Figure 5.1?
a. | hexose | b. | fructose | c. | glucose | d. | A and B
only | e. | A and C only |
|
|
|
17.
|
Lactose, a sugar in milk, is composed of one
glucose molecule joined by a glycosidic linkage to one galactose molecule. How is lactose
classified?
a. | as a pentose | b. | as a hexose | c. | as a
monosaccharide | d. | as a
disaccharide | e. | as a
polysaccharide |
|
|
|
18.
|
All of the following are polysaccharides
except
a. | lactose | b. | starch | c. | chitin | d. | cellulose | e. | amylopectin |
|
|
|
19.
|
Which of the following is true of both
starch and cellulose?
a. | They are both polymers of
glucose. | b. | They are geometric isomers of each
other. | c. | They can both be digested by
humans. | d. | They are both used for energy storage in
plants. | e. | They are both structural components of the plant cell
wall. |
|
|
|
20.
|
Which of the following is true of
cellulose?
a. | It is a polymer composed of sucrose
monomers. | b. | It is a storage polysaccharide for energy in plant
cells. | c. | It is a storage polysaccharide for energy in animal
cells. | d. | It is a major structural component of plant cell
walls. | e. | It is a major structural component of animal cell plasma
membranes. |
|
|
|
21.
|
Humans can digest starch but not cellulose
because
a. | the monomer of starch is glucose, while the monomer of
cellulose is galactose. | b. | humans have
enzymes that can hydrolyze the beta (â) glycosidic linkages of starch but not the alpha (á)
glycosidic linkages of cellulose. | c. | humans have
enzymes that can hydrolyze the alpha (á) glycosidic linkages of starch but not the beta (â)
glycosidic linkages of cellulose. | d. | humans harbor
starch-digesting bacteria in the digestive tract. | e. | the monomer of starch is glucose, while the monomer of cellulose is
maltose. |
|
|
|
22.
|
All of the following statements concerning
saturated fats are true except
a. | They are more common in animals than in
plants. | b. | They have multiple double bonds in the
carbon chains of their fatty acids. | c. | They generally
solidify at room temperature. | d. | They contain more
hydrogen than unsaturated fats having the same number of carbon atoms. | e. | They are one of several factors that contribute to
atherosclerosis. |
|
|
|
23.
|
A molecule with the formula 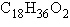 is probably a
is probably a
a. | carbohydrate. | b. | fatty acid. | c. | protein. | d. | nucleic
acid. | e. | hydrocarbon. |
|
|
|
24.
|
Which of the following statements is false
for the class of biological molecules known as lipids?
a. | They are soluble in water. | b. | They are an important constituent of cell
membranes. | c. | They contain more
energy than proteins and carbohydrates. | d. | They are not true
polymers. | e. | They contain waxes and
steroids. |
|
|
|
25.
|
What is a triacylglycerol?
a. | a protein with tertiary
structure | b. | a lipid made with three fatty acids and
glycerol | c. | a lipid that makes up much of the plasma
membrane | d. | a molecule formed from three alcohols by dehydration
reactions | e. | a carbohydrate with three sugars joined together by
glycosidic linkages |
|
|
|
26.
|
Which of the following is true regarding
saturated fatty acids?
a. | They are the predominant fatty acid in corn
oil. | b. | They have double bonds between carbon atoms of the fatty
acids. | c. | They are the principal molecules in lard and
butter. | d. | They are usually liquid at room
temperature. | e. | They are usually
produced by plants. |
|
|
|
27.
|
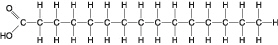
Figure
5.2
Which of the following statements is true regarding the molecule illustrated in Figure
5.2?a. | It is a saturated fatty acid. | b. | A diet rich in this molecule may contribute to
atherosclerosis. | c. | Molecules of this
type are usually liquid at room temperature. | d. | A and B
only | e. | A, B and C |
|
|
|
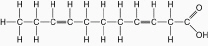
Figure
5.3
|
|
|
28.
|
Which of the following statements is true regarding
the molecule illustrated in Figure 5.3?
a. | It is a saturated fatty acid. | b. | A diet rich in this molecule may contribute to
atherosclerosis. | c. | Molecules of this
type are usually liquid at room temperature. | d. | A and B
only | e. | A, B and C |
|
|
|
29.
|
The molecule shown in Figure 5.3 is a
a. | polysaccharide. | b. | polypeptide. | c. | saturated fatty
acid. | d. | triacylglycerol. | e. | unsaturated fatty acid. |
|
|
|
30.
|
Large organic molecules are usually assembled by
polymerization of a few kinds of simple subunits. Which of the following is an exception to
this statement?
a. | a steroid | b. | cellulose | c. | DNA | d. | an
enzyme | e. | a contractile
protein |
|
|
|
31.
|
The hydrogenation of vegetable oil results in which
of the following?
a. | saturated fats and unsaturated fats with trans
double bonds | b. | an increased
contribution to artherosclerosis | c. | the oil (fat)
being a solid at room temperature | d. | A and C
only | e. | A, B, and C |
|
|
|
32.
|
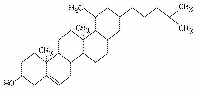
Figure
5.4
What is the structure shown in Figure 5.4?a. | starch molecule | b. | protein molecule | c. | steroid
molecule | d. | cellulose molecule | e. | phospholipid molecule |
|
|
|
33.
|
Why are human sex hormones considered to be
lipids?
a. | They are essential components of cell
membranes. | b. | They are steroids,
which are not soluble in water. | c. | They are made of
fatty acids. | d. | They are
hydrophilic compounds. | e. | They contribute to
atherosclerosis. |
|
|
|
34.
|
All of the following contain amino acids
except
a. | hemoglobin. | b. | cholesterol. | c. | antibodies. | d. | enzymes. | e. | insulin. |
|
|
|
35.
|
The bonding of two amino acid molecules to form a
larger molecule requires
a. | the release of a water
molecule. | b. | the release of a carbon dioxide
molecule. | c. | the addition of a nitrogen
atom. | d. | the addition of a water
molecule. | e. | both B and C |
|
|
|
36.
|
There are 20 different amino acids. What makes one
amino acid different from another?
a. | different carboxyl groups attached to an alpha (á)
carbon | b. | different amino groups attached to an alpha (á)
carbon | c. | different side chains (R groups) attached to an alpha
(á) carbon | d. | different alpha
(á) carbons | e. | different
asymmetric carbons |
|
|
|
37.
|
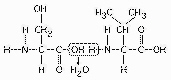
Figure
5.5
Which of the following statements is/are true regarding the chemical reaction illustrated
in Figure 5.5?a. | It is a hydrolysis reaction. | b. | It results in a peptide bond. | c. | It joins two fatty acids together. | d. | A and B only | e. | A, B, and
C |
|
|
|
38.
|
The bonding of two amino acid molecules to form a
larger molecule requires which of the following?
a. | removal of a water molecule | b. | addition of a water molecule | c. | formation of an ionic bond | d. | formation of a hydrogen bond | e. | both A and C |
|
|
|
39.
|
Polysaccharides, lipids, and proteins are similar
in that they
a. | are synthesized from monomers by the process of
hydrolysis. | b. | are synthesized
from monomers by dehydration reactions. | c. | are synthesized as
a result of peptide bond formation between monomers. | d. | are decomposed into their subunits by dehydration
reactions. | e. | all contain
nitrogen in their monomer building blocks. |
|
|
|
40.
|
Dehydration reactions are used in forming which of
the following compounds?
a. | triacylglycerides | b. | polysaccharides | c. | proteins | d. | A and C
only | e. | A, B, and C |
|
|
|
41.
|
Upon chemical analysis, a particular polypeptide
was found to contain 100 amino acids. How many peptide bonds are present in this
protein?
|
|
|
42.
|
How many different kinds of polypeptides, each
composed of 12 amino acids, could be synthesized using the 20 common amino acids?
|
|
|
43.
|
Which bonds are created during the formation of the
primary structure of a protein?
a. | peptide bonds | b. | hydrogen bonds | c. | disulfide
bonds | d. | phosphodiester bonds | e. | A, B, and C |
|
|
|
44.
|
What maintains the secondary structure of a
protein?
a. | peptide bonds | b. | hydrogen bonds | c. | disulfide
bonds | d. | ionic bonds | e. | phosphodiester bonds |
|
|
|
45.
|
Which type of interaction stabilizes the alpha
(á) helix and the beta (â) pleated sheet structures of proteins?
a. | hydrophobic interactions | b. | nonpolar covalent bonds | c. | ionic
bonds | d. | hydrogen bonds | e. | peptide bonds |
|
|
|
46.
|
The á helix and the â pleated sheet are
both common polypeptide forms found in which level of protein structure?
a. | primary | b. | secondary | c. | tertiary | d. | quaternary | e. | all of the
above |
|
|
|

Figure
5.7
|
|
|
47.
|
The structure depicted in Figure 5.7 shows
the
a. | 1-4 linkage of the á glucose monomers of
starch. | b. | 1-4 linkage of the â glucose monomers of
cellulose. | c. | double helical
structure of a DNA molecule. | d. | á helix
secondary structure of a polypeptide. | e. | â pleated
sheet secondary structure of a polypeptide. |
|
|
|
48.
|
Figure 5.7 best illustrates the
a. | secondary structure of a
polypeptide. | b. | tertiary structure
of a polypeptide. | c. | quaternary
structure of a protein. | d. | double helix
structure of DNA. | e. | primary structure
of a polysaccharide. |
|
|
|
49.
|
The tertiary structure of a protein is
the
a. | bonding together of several polypeptide chains by weak
bonds. | b. | order in which amino acids are joined in a polypeptide
chain. | c. | unique three-dimensional shape of the fully folded
polypeptide. | d. | organization of a
polypeptide chain into an á helix or â pleated sheet. | e. | overall protein structure resulting from the aggregation of two or more
polypeptide subunits. |
|
|
|
50.
|
A strong covalent bond between amino acids that
functions in maintaining a polypeptide's specific three-dimensional shape is a
(an)
a. | ionic bond. | b. | hydrophobic interaction. | c. | van der Waals
interaction. | d. | disulfide
bond. | e. | hydrogen bond. |
|
|
|
51.
|
At which level of protein structure are
interactions between the side chains (R groups) most important?
a. | primary | b. | secondary | c. | tertiary | d. | quaternary | e. | all of the
above |
|
|
|
52.
|
The R group or side chain of the amino acid serine
is – 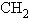 –OH. The R group or side chain of the
amino acid alanine is – –OH. The R group or side chain of the
amino acid alanine is – 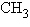 . Where would you expect to find these amino
acids in a globular protein in aqueous solution? . Where would you expect to find these amino
acids in a globular protein in aqueous solution?
a. | Serine would be in the interior, and alanine would be on
the exterior of the globular protein. | b. | Alanine would be
in the interior, and serine would be on the exterior of the globular
protein. | c. | Both serine and alanine would be in the interior of the
globular protein. | d. | Both serine and
alanine would be on the exterior of the globular protein. | e. | Both serine and alanine would be in the interior and on the exterior of the
globular protein. |
|
|
|
53.
|
Misfolding of polypeptides is a serious problem in
cells. Which of the following diseases are associated with an accumulation of misfolded
proteins?
a. | Alzheimer's | b. | Parkinson's | c. | diabetes | d. | A and B
only | e. | A, B, and C |
|
|
|
54.
|
What would be an unexpected consequence of changing
one amino acid in a protein consisting of 325 amino acids?
a. | The primary structure of the protein would be
changed. | b. | The tertiary structure of the protein might be
changed. | c. | The biological activity or function of the protein might
be altered. | d. | Only A and C are
correct. | e. | A, B, and C are
correct. |
|
|
|
55.
|
Altering which of the following levels of
structural organization could change the function of a protein?
a. | primary | b. | secondary | c. | tertiary | d. | quaternary | e. | all of the
above |
|
|
|
56.
|
What method did Frederick Sanger use to elucidate
the structure of insulin?
a. | X-ray crystallography | b. | bioinformatics | c. | analysis of amino
acid sequence of small fragments | d. | NMR
spectroscopy | e. | high-speed
centrifugation |
|
|
|
57.
|
Roger Kornberg used this method for elucidating the
structure of RNA polymerase.
a. | X-ray crystallography | b. | bioinformatics | c. | analysis of amino
acid sequence of small fragments | d. | NMR
spectroscopy | e. | high-speed
centrifugation |
|
|
|
58.
|
Which of the following uses the amino acid
sequences of polypeptides to predict a protein's three-dimensional structure?
a. | X-ray crystallography | b. | bioinformatics | c. | analysis of amino
acid sequence of small fragments | d. | NMR
spectroscopy | e. | high-speed
centrifugation |
|
|
|
59.
|
The function of each protein is a consequence of
its specific shape. What is the term used for a change in a protein's three-dimensional shape or
conformation due to disruption of hydrogen bonds, disulfide bridges, or ionic bonds?
a. | hydrolysis | b. | stabilization | c. | destabilization | d. | renaturation | e. | denaturation |
|
|
|
60.
|
What is the term used for a protein molecule that
assists in the proper folding of other proteins?
a. | tertiary protein | b. | chaperonin | c. | enzyme
protein | d. | renaturing protein | e. | denaturing protein |
|
|
|
61.
|
DNAase is an enzyme that catalyzes the hydrolysis
of the covalent bonds that join nucleotides together. What would first happen to DNA molecules
treated with DNAase?
a. | The two strands of the double helix would
separate. | b. | The phosphodiester bonds between deoxyribose sugars
would be broken. | c. | The purines would
be separated from the deoxyribose sugars. | d. | The pyrimidines
would be separated from the deoxyribose sugars. | e. | All bases would be
separated from the deoxyribose sugars. |
|
|
|
62.
|
Which of the following statements about the 5'
end of a polynucleotide strand of DNA is correct?
a. | The 5' end has a hydroxyl group attached to the
number 5 carbon of ribose. | b. | The 5' end
has a phosphate group attached to the number 5 carbon of ribose. | c. | The 5' end has thymine attached to the number 5 carbon of
ribose. | d. | The 5' end has a carboxyl group attached to the
number 5 carbon of ribose. | e. | The 5' end is
the fifth position on one of the nitrogenous bases. |
|
|
|
63.
|
Of the following functions, the major purpose of
RNA is to
a. | transmit genetic information to
offspring. | b. | function in the
synthesis of protein. | c. | make a copy of
itself, thus ensuring genetic continuity. | d. | act as a pattern
or blueprint to form DNA. | e. | form the genes of
higher organisms. |
|
|
|
64.
|
Which of the following best describes the
flow of information in eukaryotic cells?
|
|
|
65.
|
Which of the following descriptions best
fits the class of molecules known as nucleotides?
a. | a nitrogenous base and a phosphate
group | b. | a nitrogenous base and a pentose
sugar | c. | a nitrogenous base, a phosphate group, and a pentose
sugar | d. | a phosphate group and an adenine or
uracil | e. | a pentose sugar and a purine or
pyrimidine |
|
|
|
66.
|
Which of the following are nitrogenous bases of the
pyrimidine type?
a. | guanine and adenine | b. | cytosine and uracil | c. | thymine and
guanine | d. | ribose and deoxyribose | e. | adenine and thymine |
|
|
|
67.
|
Which of the following are nitrogenous bases of the
purine type?
a. | cytosine and guanine | b. | guanine and adenine | c. | adenine and
thymine | d. | thymine and uracil | e. | uracil and cytosine |
|
|
|
68.
|
If a DNA sample were composed of 10% thymine, what
would be the percentage of guanine?
a. | 10 | b. | 20 | c. | 40 | d. | 80 | e. | impossible to tell
from the information given |
|
|
|
69.
|
A double-stranded DNA molecule contains a total of
120 purines and 120 pyrimidines. This DNA molecule could be composed of
a. | 120 adenine and 120 uracil
molecules. | b. | 120 thymine and
120 adenine molecules. | c. | 120 cytosine and
120 thymine molecules. | d. | 240 adenine and
240 cytosine molecules. | e. | 240 guanine and
240 thymine molecules. |
|
|
|
70.
|
The difference between the sugar in DNA and the
sugar in RNA is that the sugar in DNA
a. | is a six-carbon sugar and the sugar in RNA is a
five-carbon sugar. | b. | can form a
double-stranded molecule. | c. | has a six-membered
ring of carbon and nitrogen atoms. | d. | can attach to a
phosphate. | e. | contains one less
oxygen atom. |
|
|
|
71.
|
Which of the following statements best
summarizes the structural differences between DNA and RNA?
a. | RNA is a protein, whereas DNA is a nucleic
acid. | b. | DNA is a protein, whereas RNA is a nucleic
acid. | c. | DNA nucleotides contain a different sugar than RNA
nucleotides. | d. | RNA is a double
helix, but DNA is single-stranded. | e. | A and D are
correct. |
|
|
|
72.
|
In the double helix structure of nucleic acids,
cytosine hydrogen bonds to
a. | deoxyribose. | b. | ribose. | c. | adenine. | d. | thymine. | e. | guanine. |
|
|
|
73.
|
If one strand of a DNA molecule has the sequence of
bases 5'ATTGCA3', the other complementary strand would have the sequence
a. | 5'TAACGT3'. | b. | 3'TAACGT5'. | c. | 5'UAACGU3'. | d. | 3'UAACGU5'. | e. | 5'UGCAAU3'. |
|
|
|
74.
|
What is the structural feature that allows DNA to
replicate?
a. | sugar-phosphate backbone | b. | complementary pairing of the nitrogenous bases | c. | disulfide bonding (bridging) of the two helixes | d. | twisting of the molecule to form an á helix | e. | three-component structure of the
nucleotides |
|
|
|
75.
|
A new organism is discovered in the forests of
Costa Rica. Scientists there determine that the polypeptide sequence of hemoglobin from the new
organism has 72 amino acid differences from humans, 65 differences from a gibbon, 49 differences from
a rat, and 5 differences from a frog. These data suggest that the new organism
a. | is more closely related to humans than to
frogs. | b. | is more closely related to frogs than to
humans. | c. | may have evolved from gibbons but not
rats. | d. | is more closely related to humans than to
rats. | e. | may have evolved from rats but not from humans and
gibbons. |
|
|
|
76.
|
Which of the following is an example of
hydrolysis?
a. | the reaction of two monosaccharides, forming a
disaccharide with the release of water | b. | the synthesis of
two amino acids, forming a peptide with the release of water | c. | the reaction of a fat, forming glycerol and fatty acids with the release of
water | d. | the reaction of a fat, forming glycerol and fatty acids
with the utilization of water | e. | the synthesis of a
nucleotide from a phosphate, a pentose sugar, and a nitrogenous base with the production of a
molecule of water |
|
|
|
77.
|
The element nitrogen is present in all of the
following except
a. | proteins. | b. | nucleic acids. | c. | amino
acids. | d. | DNA. | e. | monosaccharides. |
|
|
|
78.
|
Which of the following is a diverse group of
hydrophobic molecules?
a. | carbohydrates | b. | lipids | c. | proteins | d. | nucleic
acids |
|
|
|
79.
|
Which of the following store and transmit
hereditary information?
a. | carbohydrates | b. | lipids | c. | proteins | d. | nucleic
acids |
|
|
|
80.
|
Enzymes are
a. | carbohydrates. | b. | lipids. | c. | proteins. | d. | nucleic
acids. |
|
|
|
The following questions are based on the 15
molecules illustrated in Figure 5.8. Each molecule may be used once, more than once, or not at
all.
Figure 5.8
|
|
|
81.
|
Which molecule has hydrophilic and hydrophobic
properties and would be found in plasma membranes?
|
|
|
82.
|
Which of the following combinations could be linked
together to form a nucleotide?
a. | 1, 2, and 11 | b. | 3, 7, and 8 | c. | 5, 9, and
10 | d. | 11, 12, and 13 | e. | 12, 14, and 15 |
|
|
|
83.
|
Which of the following molecules contain(s) an
aldehyde type of carbonyl functional group?
|
|
|
84.
|
Which molecule is glycerol?
|
|
|
85.
|
Which molecule is a saturated fatty
acid?
|
|
|
86.
|
Which of the following molecules is a purine type
of nitrogenous base?
|
|
|
87.
|
Which of the following molecules act as building
blocks (monomers) of polypeptides?
a. | 1, 4, and 6 | b. | 2, 7, and 8 | c. | 7, 8, and
13 | d. | 11, 12, and 13 | e. | 12, 13, and 15 |
|
|
|
88.
|
Which of the following molecules is an amino acid
with a hydrophobic R group or side chain?
|
|
|
89.
|
Which of the following molecules could be joined
together by a peptide bond as a result of a dehydration reaction?
a. | 2 and 3 | b. | 3 and 7 | c. | 7 and
8 | d. | 8 and 9 | e. | 12 and 13 |
|
|
|
90.
|
A fat (or triacylglycerol) would be formed as a
result of a dehydration reaction between
a. | one molecule of 9 and three molecules of
10. | b. | three molecules of 9 and one molecule of
10. | c. | one molecule of 5 and three molecules of
9. | d. | three molecules of 5 and one molecule of
9. | e. | one molecule of 5 and three molecules of
10. |
|
|
|
91.
|
Which of the following molecules could be joined
together by a phosphodiester type of covalent bond?
a. | 3 and 4 | b. | 3 and 8 | c. | 6 and
15 | d. | 11 and 12 | e. | 11 and 13 |
|
|
|
92.
|
Which of the following molecules is the pentose
sugar found in RNA?
|
|
|
93.
|
Which of the following molecules contains a
glycosidic linkage type of covalent bond?
|
|
|
94.
|
Which of the following molecules has (have) a
functional group that frequently is involved in maintaining the tertiary structure of a
protein?
|
|
|
95.
|
Which of the following molecules consists of a
hydrophilic "head" region and a hydrophobic "tail" region?
|
|
|
96.
|
Which of the following statements is
false?
a. | 1 and 4 could be joined together by a glycosidic linkage
to form a disaccharide. | b. | 9 and 10 could be
joined together by ester bonds to form a triacylglycerol. | c. | 2 and 7 could be joined together to form a short
peptide. | d. | 2, 7, and 8 could be joined together to form a short
peptide. | e. | 14 and 15 could be joined together to form a
polypeptide. |
|
|
|
97.
|
Which term includes all others in the
list?
a. | monosaccharide | b. | disaccharide | c. | starch | d. | carbohydrate | e. | polysaccharide |
|
|
|
98.
|
The molecular formula for glucose is
C6H12O6. What would be the molecular formula for a polymer made by
linking ten glucose molecules together by dehydration reactions?
|
|
|
99.
|
The enzyme amylase can break glycosidic linkages
between glucose monomers only if the monomers are the á form. Which of the following could
amylase break down?
a. | glycogen, starch, and
amylopectin | b. | glycogen and
cellulose | c. | cellulose and chitin | d. | starch and chitin | e. | starch,
amylopectin, and cellulose |
|
|
|
100.
|
Which of the following statements concerning
unsaturated fats is true?
a. | They are more common in animals than in
plants. | b. | They have double bonds in the carbon chains of their
fatty acids. | c. | They generally
solidify at room temperature. | d. | They contain more
hydrogen than saturated fats having the same number of carbon atoms. | e. | They have fewer fatty acid molecules per fat
molecule. |
|
|
|
101.
|
The structural level of a protein least affected by
a disruption in hydrogen bonding is the
a. | primary level. | b. | secondary level. | c. | tertiary
level. | d. | quaternary level. | e. | All structural levels are equally
affected. |
|
|
|
102.
|
Which of the following pairs of base sequences
could form a short stretch of a normal double helix of DNA?
a. | 5'-purine-pyrimidine-purine-pyrimidine-3' with
3'-purine-pyrimidine-purine-pyrimidine-5' | b. | 5'-A-G-C-T-3' with
5'-T-C-G-A-3' | c. | 5'-G-C-G-C-3' with
5'-T-A-T-A-3' | d. | 5'-A-T-G-C-3' with
5'-G-C-A-T-3' | e. | All of these pairs
are correct. |
|
|
|
103.
|
Enzymes that break down DNA catalyze the hydrolysis
of the covalent bonds that join nucleotides together. What would happen to DNA molecules treated with
these enzymes?
a. | The two strands of the double helix would
separate. | b. | The phosphodiester linkages between deoxyribose sugars
would be broken. | c. | The purines would
be separated from the deoxyribose sugars. | d. | The pyrimidines
would be separated from the deoxyribose sugars. | e. | All bases would be
separated from the deoxyribose sugars. |
|
Short Answer
|
|
|
Refer to Figure 5.6 to answer the following
questions.
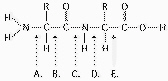
Figure 5.6
|
|
|
104.
|
At which bond would water need to be added to
achieve hydrolysis of the peptide, back to its component amino acid?
|
|
|
105.
|
Which bond is a peptide bond?
|
|
|
106.
|
Which bond is closest to the N-terminus of the
molecule?
|
|
|
107.
|
Which bond is closest to the carboxyl end of the
molecule?
|
|
|
108.
|
Construct a table that organizes the following
terms, and label the columns and rows.
| phosphodiester linkages | polypeptides | monosaccharides | | peptide bonds | triacylglycerols | nucleotides | | glycosidic linkages | polynucleotides | amino acids | | ester linkages | polysaccharides | fatty acids | | | |
|
|
|
109.
|
Draw the polynucleotide strand in Figure 5.27a from
your textbook, and label the bases G, T, C, and T, starting from the 5' end. Now, draw the
complementary strand of the double helix, using the same symbols for phosphates (circles), sugars
(pentagons), and bases. Label the bases. Draw arrows showing the 5'  3'
direction of each strand. Use the arrows to make sure the second strand is antiparallel to the first.
Hint: After you draw the first strand vertically, turn the paper upside down; it is easier to draw
the second strand from the 5' toward the 3' direction as you go from top to
bottom. 3'
direction of each strand. Use the arrows to make sure the second strand is antiparallel to the first.
Hint: After you draw the first strand vertically, turn the paper upside down; it is easier to draw
the second strand from the 5' toward the 3' direction as you go from top to
bottom.
|