Multiple Choice
Identify the
choice that best completes the statement or answers the question.
|
|
|
1.
|
About 25 of the 92 natural elements are known to be
essential to life. Which four of these 25 elements make up approximately 96% of living
matter?
a. | carbon, sodium, chlorine,
nitrogen | b. | carbon, sulfur, phosphorus,
hydrogen | c. | oxygen, hydrogen, calcium,
sodium | d. | carbon, hydrogen, nitrogen,
oxygen | e. | carbon, oxygen, sulfur,
calcium |
|
|
|
2.
|
Trace elements are those required by an organism in
only minute quantities. Which of the following is a trace element that is required by humans and
other vertebrates?
a. | nitrogen | b. | calcium | c. | iodine | d. | sodium | e. | phosphorus |
|
|
|
3.
|
Three or four of the following statements are true
and correct. Which one, if any, is false?
a. | Carbon, hydrogen, oxygen, and nitrogen make up
approximately 96% of living matter. | b. | The trace element
iodine is required only in very small quantities by vertebrates. | c. | Virtually all organisms require the same elements in the same
quantities. | d. | Iron is an example
of an element needed by all organisms. |
|
|
|
4.
|
Which of the following statements is
false?
a. | Atoms of the various elements differ in their number of
subatomic particles. | b. | All atoms of a
particular element have the same number of protons in their nuclei. | c. | The neutrons and protons present in the nucleus of an atom are almost
identical in mass; each has a mass of about 1 dalton. | d. | An atom is the smallest unit of an element that still retains the properties
of the element. | e. | Protons and
electrons are electrically charged particles. Protons have one unit of negative charge, and electrons
have one unit of positive charge. |
|
|
|
5.
|
Each element is unique and different from other
elements because of the number of protons in the nuclei of its atoms. Which of the following
indicates the number of protons in an atom's nucleus?
a. | atomic mass | b. | atomic weight | c. | atomic
number | d. | mass weight | e. | mass number |
|
|
|
6.
|
The mass number of an element can be easily
approximated by adding together the number of ____ in an atom of that element.
a. | protons and neutrons | b. | energy levels | c. | protons and
electrons | d. | neutrons and electrons | e. | isotopes |
|
|
|
7.
|
What is the approximate atomic mass of an atom with
16 neutrons, 15 protons, and 15 electrons?
a. | 15 daltons | b. | 16 daltons | c. | 30
daltons | d. | 31 daltons | e. | 46 daltons |
|
|
|
8.
|
Oxygen has an atomic number of 8 and a mass number
of 16. Thus, the atomic mass of an oxygen atom is
a. | exactly 8 grams. | b. | exactly 8 daltons. | c. | approximately 16
grams. | d. | approximately 16 daltons. | e. | 24 amu (atomic mass units). |
|
|
|
9.
|
The nucleus of a nitrogen atom contains 7 neutrons
and 7 protons. Which of the following is a correct statement concerning
nitrogen?
a. | The nitrogen atom has a mass number of approximately 7
daltons and an atomic mass of 14. | b. | The nitrogen atom
has a mass number of approximately 14 daltons and an atomic mass of 7. | c. | The nitrogen atom has a mass number of 14 and an atomic mass of 7
grams. | d. | The nitrogen atom has a mass number of 7 grams and an
atomic number of 14. | e. | The nitrogen atom
has a mass number of 14 and an atomic mass of approximately 14
daltons. |
|
|
|
10.
|
Calcium has an atomic number of 20 and an atomic
mass of 40. Therefore, a calcium atom must have
a. | 20 protons. | b. | 40 electrons. | c. | 40
neutrons. | d. | A and B only | e. | A, B, and C |
|
|
|
11.
|
An atom with an atomic number of 9 and a mass
number of 19 would have an atomic mass of approximately
a. | 9 daltons. | b. | 9 grams. | c. | 10
daltons. | d. | 20 grams. | e. | 19 daltons. |
|
|
|
12.
|
Different atomic forms of an element contain the
same number of protons but a different number of neutrons. What are these different atomic forms
called?
a. | ions | b. | isotopes | c. | neutronic
atoms | d. | isomers | e. | radioactive atoms |
|
|
|
13.
|
How do isotopes of the same element differ from
each other?
a. | number of protons | b. | number of electrons | c. | number of
neutrons | d. | valence electron distribution | e. | amount of radioactivity |
|
|
|
14.
|
Which of the following best describes the
relationship between the atoms described below?
a. | They are isomers. | b. | They are polymers. | c. | They are
isotopes. | d. | They contain 1 and 3 protons,
respectively. | e. | They each contain
1 neutron. |
|
|
|
15.
|
Which of the following best describes the
relationship between the atoms described below?
a. | They contain 31 and 32 electrons,
respectively. | b. | They are both
phosphorus cations. | c. | They are both
phosphorus anions. | d. | They are both
isotopes of phosphorus. | e. | They contain 31
and 32 protons, respectively. |
|
|
|
16.
|
One difference between carbon-12 (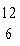 C) and carbon-14 ( C) and carbon-14 (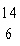 C) is that carbon-14 has C) is that carbon-14 has
a. | two more protons than
carbon-12. | b. | two more electrons
than carbon-12. | c. | two more neutrons
than carbon-12. | d. | A and C
only | e. | B and C only |
|
|
|
17.
|
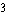 H is a radioactive isotope
of hydrogen. One difference between hydrogen-1 ( H is a radioactive isotope
of hydrogen. One difference between hydrogen-1 (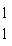 H) and hydrogen-3 ( H) and hydrogen-3 (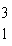 H) is that hydrogen-3 has H) is that hydrogen-3 hasa. | one more neutron and one more proton than
hydrogen-1. | b. | one more proton
and one more electron than hydrogen-1. | c. | one more electron
and one more neutron than hydrogen-1. | d. | two more neutrons
than hydrogen-1. | e. | two more protons
than hydrogen-1. |
|
|
|
18.
|
The atomic number of carbon is 6. Carbon-14 is
heavier than carbon-12 because the atomic nucleus of carbon-14 contains ____ neutrons.
|
|
|
19.
|
Electrons exist only at fixed levels of potential
energy. However, if an atom absorbs sufficient energy, a possible result is that
a. | an electron may move to an electron shell farther out
from the nucleus. | b. | an electron may
move to an electron shell closer to the nucleus. | c. | the atom may become a radioactive isotope. | d. | the atom would become a positively charged ion, or
cation. | e. | the atom would become a negatively charged ion, or
anion. |
|
|
|
20.
|
The atomic number of neon is 10. Therefore, which
of the following is correct about an atom of neon?
a. | It has 8 electrons in its outer electron
shell. | b. | It is inert. | c. | It has an atomic mass of 10 daltons. | d. | A and B only | e. | A, B, and C are
correct. |
|
|
|
21.
|
From its atomic number of 15, it is possible to
predict that the phosphorus atom has
a. | 15 neutrons. | b. | 15 protons. | c. | 15
electrons. | d. | 8 electrons in its
outermost electron shell. | e. | B and C
only |
|
|
|
22.
|
Atoms whose outer electron shells contain eight
electrons tend to
a. | form ionic bonds in aqueous
solutions. | b. | form covalent
bonds in aqueous solutions. | c. | be stable and
chemically nonreactive, or inert. | d. | be unstable and
chemically very reactive. | e. | be isotopes and
very radioactive. |
|
|
|
Use the information extracted from the periodic
table in Figure 2.2 to answer the following questions.
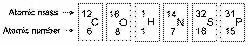
Figure 2.2
|
|
|
23.
|
How many electrons does nitrogen have in its
valence shell?
|
|
|
24.
|
How many electrons does phosphorus have in its
valence shell?
|
|
|
25.
|
How many neutrons are present in the nucleus of a
phosphorus atom?
|
|
|
26.
|
How many electrons does an atom of sulfur have in
its valence shell?
|
|
|
27.
|
Based on electron configuration, which of these
elements would exhibit chemical behavior most like that of oxygen?
a. | carbon | b. | hydrogen | c. | nitrogen | d. | sulfur | e. | phosphorus |
|
|
|
28.
|
How many electrons would be expected in the
outermost electron shell of an atom with atomic number 12?
|
|
|
29.
|
The atomic number of each atom is given to the left
of each of the elements below. Which of the atoms has the same valence as carbon (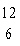 C)? C)?
a. | 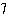 nitrogen nitrogen | b. | 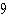 flourine flourine | c. | 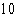 neon neon | d. | 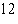 magnesium magnesium | e. | 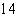 silicon silicon |
|
|
|
30.
|
What is the valence of an atom with six electrons
in its outer electron shell?
|
|
|
31.
|
Fluorine has an atomic number of 9 and a mass
number of 19. How many electrons are needed to complete the valence shell of a fluorine
atom?
|
|
|
32.
|
What is the maximum number of electrons in the
1s orbital of an atom?
|
|
|
33.
|
What is the maximum number of electrons in a 2p
orbital of an atom?
|
|
|
34.
|
A covalent chemical bond is one in
which
a. | electrons are removed from one atom and transferred to
another atom so that the two atoms become oppositely charged. | b. | protons and neutrons are shared by two atoms so as to satisfy the requirements
of both atoms. | c. | outer-shell
electrons of two atoms are shared so as to satisfactorily fill the outer electron shells of both
atoms. | d. | outer-shell electrons of one atom are transferred to the
inner electron shells of another atom. | e. | the inner-shell
electrons of one atom are transferred to the outer shell of another
atom. |
|
|
|
35.
|
If an atom of sulfur (atomic number 16) were
allowed to react with atoms of hydrogen (atomic number 1), which of the molecules below would be
formed?
a. | S – H | b. | H – S – H | c. | 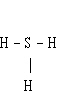 | d. | 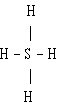 | e. | H = S =
H |
|
|
|
36.
|
What is the maximum number of covalent bonds an
element with atomic number 8 can make with hydrogen?
|
|
|
37.
|
A molecule of carbon dioxide (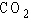 )
is formed when one atom of carbon (atomic number 6) is covalently bonded with two atoms of oxygen
(atomic number 8). What is the total number of electrons that must be shared between the carbon atom
and the oxygen atoms in order to complete the outer electron shell of all three atoms? )
is formed when one atom of carbon (atomic number 6) is covalently bonded with two atoms of oxygen
(atomic number 8). What is the total number of electrons that must be shared between the carbon atom
and the oxygen atoms in order to complete the outer electron shell of all three atoms?
|
|
|
38.
|
Nitrogen (N) is much more electronegative than
hydrogen (H). Which of the following statements is correct about the atoms in ammonia (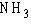 )? )?
a. | Each hydrogen atom has a partial positive
charge. | b. | The nitrogen atom has a strong positive
charge. | c. | Each hydrogen atom has a slight negative
charge. | d. | The nitrogen atom has a partial positive
charge. | e. | There are covalent bonds between the hydrogen
atoms. |
|
|
|
39.
|
When two atoms are equally electronegative, they
will interact to form
a. | equal numbers of isotopes. | b. | ions. | c. | polar covalent
bonds. | d. | nonpolar covalent bonds. | e. | ionic bonds. |
|
|
|
40.
|
What results from an unequal sharing of electrons
between atoms?
a. | a nonpolar covalent bond | b. | a polar covalent bond | c. | an ionic
bond | d. | a hydrogen bond | e. | a hydrophobic interaction |
|
|
|
41.
|
A covalent bond is likely to be polar
when
a. | one of the atoms sharing electrons is much more
electronegative than the other atom. | b. | the two atoms
sharing electrons are equally electronegative. | c. | the two atoms
sharing electrons are of the same element. | d. | it is between two
atoms that are both very strong electron acceptors. | e. | the two atoms sharing electrons are different
elements. |
|
|
|
42.
|
Which of the following molecules contains the
strongest polar covalent bond?
|
|
|
The following questions refer to Figure
2.3.

Figure 2.3
|
|
|
43.
|
What results from the chemical reaction illustrated
in Figure 2.3?
a. | a cation with a net charge of
+1 | b. | a cation with a net charge of
–1 | c. | an anion with a net charge of
+1 | d. | an anion with a net charge of
–1 | e. | A and D |
|
|
|
44.
|
What is the atomic number of the cation formed in
the reaction illustrated in Figure 2.3?
|
|
|
45.
|
The ionic bond of sodium chloride is formed
when
a. | chlorine gains an electron from
sodium. | b. | sodium and chlorine share an electron
pair. | c. | sodium and chlorine both lose electrons from their outer
valence shells. | d. | sodium gains an
electron from chlorine. | e. | chlorine gains a
proton from sodium. |
|
|
|
46.
|
What is the difference between covalent bonds and
ionic bonds?
a. | Covalent bonds involve the sharing of protons between
atoms, and ionic bonds involve the sharing of electrons between atoms. | b. | Covalent bonds involve the sharing of neutrons between atoms, and ionic bonds
involve the sharing of electrons between atoms. | c. | Covalent bonds
involve the sharing of electrons between atoms, and ionic bonds involve the electrical attraction
between atoms. | d. | Covalent bonds
involve the sharing of protons between atoms, and ionic bonds involve the sharing of neutrons between
atoms. | e. | Covalent bonds involve the transfer of electrons between
atoms, and ionic bonds involve the sharing of neutrons between
atoms. |
|
|
|
47.
|
In ammonium chloride salt (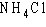 )
the anion is a single chloride ion, )
the anion is a single chloride ion, 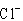 . What is the cation of . What is the cation of 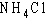 ? ?
a. | N, with a charge of +3 | b. | H, with a charge of +1 | c. | H2 with a charge
of +4 | d. | NH4 with a charge of +1 | e. | NH4 with a charge of +4 |
|
|
|
48.
|
The atomic number of chlorine is 17. The atomic
number of magnesium is 12. What is the formula for magnesium chloride?
a. | MgCl | b. | MgCl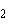 | c. | Mg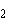 Cl Cl | d. | Mg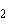 Cl Cl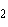 | e. | MgCl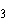 |
|
|
|
49.
|
Which of the following results from a transfer of
electron(s) between atoms?
a. | nonpolar covalent bond | b. | polar covalent bond | c. | ionic
bond | d. | hydrogen bond | e. | hydrophobic interaction |
|
|
|
50.
|
Which of the following explains most specifically
the attraction of water molecules to one another?
a. | nonpolar covalent bond | b. | polar covalent bond | c. | ionic
bond | d. | hydrogen bond | e. | hydrophobic interaction |
|
|
|
51.
|
Van der Waals interactions result
when
a. | hybrid orbitals overlap. | b. | electrons are not symmetrically distributed in a
molecule. | c. | molecules held by ionic bonds react with
water. | d. | two polar covalent bonds
react. | e. | a hydrogen atom loses an
electron. |
|
|
|
52.
|
A van der Waals interaction is the weak attraction
between
a. | the electrons of one molecule and the electrons of a
nearby molecule. | b. | the nucleus of one
molecule and the electrons of a nearby molecule. | c. | a polar molecule and a nearby nonpolar molecule. | d. | a polar molecule and a nearby molecule that is also
polar. | e. | a nonpolar molecule and a nearby molecule that is also
nonpolar. |
|
|
|
53.
|
Which of the following is not considered to
be a weak molecular interaction?
a. | a covalent bond | b. | a van der Waals interaction | c. | an ionic bond in the presence of water | d. | a hydrogen bond | e. | A and B
only |
|
|
|
54.
|
Which of the following would be regarded as
compounds?
a. | 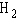 | b. | 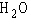 | c. | 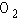 | d. | 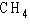 | e. | B and D, but not A and C |
|
|
|
55.
|
Sometimes atoms form molecules by sharing two pairs
of valence electrons. When this occurs, the atoms are said to be joined by
a. | a double covalent bond. | b. | an electronegative bond. | c. | a hydrogen
bond. | d. | a protonic bond. | e. | a complex bond. |
|
|
|
Refer to the following figure to answer the
following questions.
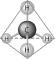
|
|
|
56.
|
The molecule shown here could be described in
chemical symbols as
|
|
|
57.
|
The molecule shown here is the simplest of organic
compounds. It is called
a. | a carbohydrate. | b. | carbon dioxide. | c. | methane. | d. | carbonic
hydrate. | e. | methyl carbonate. |
|
|
|
Refer to the following figure to answer the
following questions.
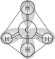
|
|
|
58.
|
In the methane molecule shown here, bonds have
formed that include both the s orbital valence electrons of the hydrogen atoms and the
p orbital valence electrons of the carbon. The electrons in these bonds are said to
have
a. | double orbitals. | b. | tetrahedral orbitals. | c. | complex
orbitals. | d. | hybrid orbitals. | e. | reduced orbitals. |
|
|
|
59.
|
Which one of the atoms shown would be most likely
to form a cation with a charge of +1?
|
|
|
60.
|
Which one of the atoms shown would be most likely
to form an anion with a charge of -1?
|
|
|
61.
|
Which of the following pairs of atoms would be most
likely to form a covalent bond?
|
|
|
62.
|
Which of the following pairs of atoms would be most
likely to form an ionic bond?
|
|
|
63.
|
The hybrid orbitals in a molecule of methane are
oriented
a. | toward the corners of a tetrahedron centered on the
carbon atom. | b. | toward the corners
of a cube centered on the carbon atom. | c. | toward the corners
of a triangle centered on the carbon atom. | d. | toward the corners
of a rectangle centered on the carbon atom. | e. | toward the edges
of an oval centered on the carbon atom. |
|
|
|
64.
|
Which of the following is true for this reaction?
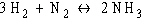
a. | The reaction is
nonreversible. | b. | Hydrogen and
nitrogen are the reactants of the reverse reaction. | c. | Hydrogen and nitrogen are the products of the forward
reaction. | d. | Ammonia is being formed and
decomposed. | e. | Hydrogen and
nitrogen are being decomposed. |
|
|
|
65.
|
Which of the following best describes chemical
equilibrium?
a. | Forward and reverse reactions continue with no effect on
the concentrations of the reactants and products. | b. | Concentrations of products are higher than the concentrations of the
reactants. | c. | Forward and
reverse reactions have stopped so that the concentration of the reactants equals the concentration of
the products. | d. | Reactions stop
only when all reactants have been converted to products. | e. | There are equal concentrations of reactants and products, and the reactions
have stopped. |
|
|
|
66.
|
Which of the following describes any reaction that
has reached chemical equilibrium?
a. | The concentration of the reactants equals the
concentration of the products. | b. | The rate of the
forward reaction is equal to the rate of the reverse reaction. | c. | All of the reactants have been converted to the products of the
reaction. | d. | All of the products have been converted to the reactants
of the reaction. | e. | Both the forward
and the reverse reactions have stopped with no net effect on the concentration of the reactants and
the products. |
|
|
|
67.
|
A group of molecular biologists is trying to
synthesize a new artificial compound to mimic the effects of a known hormone that influences sexual
behavior. They have turned to you for advice. Which of the following compounds is most likely to
mimic the effects of the hormone?
a. | a compound with the same number of carbon atoms as the
hormone | b. | a compound with the same molecular mass (measured in
daltons) as the hormone | c. | a compound with
the same three-dimensional shape as part of the hormone | d. | a compound with the same number of orbital electrons as the
hormone | e. | a compound with the same number of hydrogen and nitrogen
atoms as the hormone |
|
|
|
68.
|
In the term trace element, the modifier
trace means
a. | the element is required in very small
amounts. | b. | the element can be used as a label to trace atoms
through an organism's metabolism. | c. | the element is
very rare on Earth. | d. | the element
enhances health but is not essential for the organism's long-term
survival. | e. | the element passes rapidly through the
organism. |
|
|
|
69.
|
Compared with 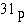 , the
radioactive isotope , the
radioactive isotope 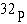 has has
a. | a different atomic number. | b. | one more neutron. | c. | one more
proton. | d. | one more electron. | e. | a different charge. |
|
|
|
70.
|
Atoms can be represented by simply listing the
number of protons, neutrons, and electrons –for example, 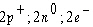 for helium.
Which one of the following lists represents the for helium.
Which one of the following lists represents the 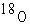 isotope of
oxygen? isotope of
oxygen?
|
|
|
71.
|
The atomic number of sulfur is 16. Sulfur combines
with hydrogen by covalent bonding to form a compound, hydrogen sulfide. Based on the number of
valence electrons in a sulfur atom, predict the molecular formula of the compound:
|
|
|
72.
|
The reactivity of an atom arises from
a. | the average distance of the outermost electron shell
from the nucleus. | b. | the existence of
unpaired electrons in the valence shell. | c. | the sum of the
potential energies of all the electron shells. | d. | the potential
energy of the valence shell. | e. | the energy
difference between the s and p orbitals. |
|
|
|
73.
|
Which statement is true of all atoms that are
anions?
a. | The atom has more electrons than
protons. | b. | The atom has more protons than
electrons. | c. | The atom has fewer
protons than does a neutral atom of the same element. | d. | The atom has more neutrons than protons. | e. | The net charge is 12. |
|
|
|
74.
|
What coefficients must be placed in the following
blanks so that all atoms are accounted for in the products?
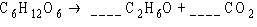
a. | 1; 2 | b. | 2; 2 | c. | 1;
3 | d. | 1; 1 | e. | 3; 1 |
|
|
|
75.
|
Which of the following statements correctly
describes any chemical reaction that has reached equilibrium?
a. | The concentrations of products and reactants are
equal. | b. | The rate of the forward reaction equals the rate of the
reverse reaction. | c. | Both forward and
reverse reactions have halted. | d. | The reaction is
now irreversible. | e. | No reactants
remain. |
|
Short Answer
|
|
|
Please refer to Figure 2.1 to answer the
following questions.

Figure 2.1
|
|
|
76.
|
Which drawing depicts the electron configuration of
neon (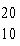 Ne)? Ne)?
|
|
|
77.
|
Which drawing depicts the electron configuration of
oxygen (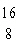 O)? O)?
|
|
|
78.
|
Which drawing depicts the electron configuration of
nitrogen (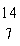 N)? N)?
|
|
|
79.
|
Which drawing is of an atom with the atomic number
of 6?
|
|
|
80.
|
Which drawing depicts an atom that is inert or
chemically unreactive?
|
|
|
81.
|
Which drawing depicts an atom with a valence of
3?
|
|
|
82.
|
Which drawing depicts an atom with a valence of
2?
|
|
|
83.
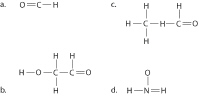
|
Draw Lewis structures for each hypothetical
molecule shown below, using the correct number of valence electrons for each atom. Determine which
molecule makes sense because each atom has a complete valence shell and each bond has the correct
number of electrons. Explain what makes the other molecules nonsensical, considering the number of
bonds each type of atom can make.
|