Multiple Choice
Identify the
choice that best completes the statement or answers the question.
|
|
|
1.
|
Which of the following statements is
false?
a. | Atoms of the various elements differ in their number of
subatomic particles. | b. | All atoms of a
particular element have the same number of protons in their nuclei. | c. | The neutrons and protons present in the nucleus of an atom are almost
identical in mass; each has a mass of about 1 dalton. | d. | An atom is the smallest unit of an element that still retains the properties
of the element. | e. | Protons and
electrons are electrically charged particles. Protons have one unit of negative charge, and electrons
have one unit of positive charge. |
|
|
|
2.
|
In a single molecule of water, two hydrogen atoms
are bonded to a single oxygen atom by
a. | hydrogen bonds. | b. | nonpolar covalent bonds. | c. | polar covalent
bonds. | d. | ionic bonds. | e. | van der Waals interactions. |
|
|
|
3.
|
An example of a hydrogen bond is the bond
between
a. | C and H in methane (CH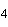 ). ). | b. | the H of one water
molecule and the O of another water molecule. | c. | Na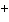 and Cl and Cl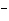 in salt. in salt. | d. | the two hydrogen atoms in a molecule of hydrogen gas (H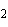 ). ). | e. | Mg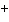 and Cl and Cl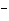 in MgCl in MgCl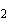 . . |
|
|
|
4.
|
Water is able to form hydrogen bonds
because
a. | oxygen has a valence of 2. | b. | the water molecule is shaped like a tetrahedron. | c. | the bonds that hold together the atoms in a water molecule are polar covalent
bonds. | d. | the oxygen atom in a water molecule has a weak positive
charge. | e. | each of the hydrogen atoms in a water molecule is weakly
negative in charge. |
|
|
|
5.
|
Carbon dioxide (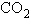 ) is readily
soluble in water, according to the equation ) is readily
soluble in water, according to the equation 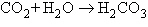 Carbonic acid ( Carbonic acid (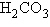 ) is a weak acid. If ) is a weak acid. If 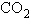 is bubbled into a beaker containing pure,
freshly-distilled water, which of the following graphs correctly describes the results? is bubbled into a beaker containing pure,
freshly-distilled water, which of the following graphs correctly describes the results?
|
|
|
6.
|
Organic chemistry is a science based on the study
of
a. | functional groups. | b. | vital forces interacting with matter. | c. | carbon compounds. | d. | water and its
interaction with other kinds of molecules. | e. | inorganic
compounds. |
|
|
|
7.
|
How many electron pairs does carbon share in order
to complete its valence shell?
|
|
|
8.
|
Which of the following statements best describes
the carbon atoms present in a seed-eating bird?
a. | They were incorporated into organic molecules by
plants. | b. | They were processed into sugars through
photosynthesis. | c. | They are
ultimately derived from carbon dioxide. | d. | Only A and C are
correct. | e. | A, B, and C are
correct. |
|
|
|
9.
|
Three or four of the following illustrations depict
different structural isomers of the organic compound with molecular formula 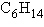 .
For clarity, only the carbon skeletons are shown; hydrogen atoms that would be attached to the
carbons have been omitted. Which one, if any, is NOT a structural isomer of this
compound? .
For clarity, only the carbon skeletons are shown; hydrogen atoms that would be attached to the
carbons have been omitted. Which one, if any, is NOT a structural isomer of this
compound?
a. | 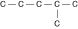 | b. | 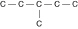 | c. | 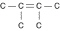 | d. | 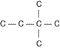 | e. | Each of the illustrations in the other answer choices depicts a structural
isomer of the compound with molecular formula 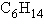 . . |
|
|
|
10.
|
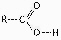
Figure
4.4
What is the name of the functional group shown in Figure 4.4?a. | carbonyl | b. | ketone | c. | aldehyde | d. | carboxyl | e. | hydroxyl |
|
|
|
11.
|
Which of the following contains nitrogen in
addition to carbon, oxygen, and hydrogen?
a. | an alcohol such as ethanol | b. | a monosaccharide such as glucose | c. | a steroid such as testosterone | d. | an amino acid such as glycine | e. | a hydrocarbon such as benzene |
|
|
|
12.
|
Which two functional groups are always found
in amino acids?
a. | ketone and aldehyde | b. | carbonyl and carboxyl | c. | carboxyl and
amino | d. | phosphate and sulfhydryl | e. | hydroxyl and aldehyde |
|
|
|
13.
|
Which of the following hydrocarbons has a double
bond in its carbon skeleton?
|
|
|
14.
|
Which of the following is not a
polymer?
a. | glucose | b. | starch | c. | cellulose | d. | chitin | e. | DNA |
|
|
|
15.
|
What is the chemical mechanism by which cells make
polymers from monomers?
a. | phosphodiester linkages | b. | hydrolysis | c. | dehydration
reactions | d. | ionic bonding of monomers | e. | the formation of disulfide bridges between
monomers |
|
|
|
16.
|
How many molecules of water are needed to
completely hydrolyze a polymer that is 11 monomers long?
|
|
|
17.
|
Which of the following best summarizes the
relationship between dehydration reactions and hydrolysis?
a. | Dehydration reactions assemble polymers, and hydrolysis
reactions break down polymers. | b. | Macromolecular
synthesis occurs through the removal of water and digestion occurs through the addition of
water. | c. | Dehydration reactions can occur only after
hydrolysis. | d. | Hydrolysis creates
monomers, and dehydration reactions break down polymers. | e. | A and B are correct. |
|
|
|
18.
|
The molecular formula for glucose is 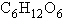 . What would be the molecular formula for a molecule made by linking three glucose
molecules together by dehydration reactions? . What would be the molecular formula for a molecule made by linking three glucose
molecules together by dehydration reactions?
|
|
|
19.
|
Lactose, a sugar in milk, is composed of one
glucose molecule joined by a glycosidic linkage to one galactose molecule. How is lactose
classified?
a. | as a pentose | b. | as a hexose | c. | as a
monosaccharide | d. | as a
disaccharide | e. | as a
polysaccharide |
|
|
|
20.
|
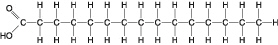
Figure
5.2
Which of the following statements is true regarding the molecule illustrated in Figure
5.2?a. | It is a saturated fatty acid. | b. | A diet rich in this molecule may contribute to
atherosclerosis. | c. | Molecules of this
type are usually liquid at room temperature. | d. | A and B
only | e. | A, B and C |
|