Multiple Choice
Identify the
choice that best completes the statement or answers the question.
|
|
|
1.
|
Organic chemistry is a science based on the study
of
a. | functional groups. | b. | vital forces interacting with matter. | c. | carbon compounds. | d. | water and its
interaction with other kinds of molecules. | e. | inorganic
compounds. |
|
|
|
2.
|
Early 19th-century scientists believed that living
organisms differed from nonliving things as a result of possessing a "life force" that
could create organic molecules from inorganic matter. The term given to this belief is
a. | organic synthesis. | b. | vitalism. | c. | mechanism. | d. | organic
evolution. | e. | inorganic
synthesis. |
|
|
|
3.
|
The experimental approach taken in current
biological investigations presumes that
a. | simple organic compounds can be synthesized in the
laboratory from inorganic precursors, but complex organic compounds like carbohydrates and proteins
can only be synthesized by living organisms. | b. | a life force
ultimately controls the activities of living organisms and this life force cannot be studied by
physical or chemical methods. | c. | although a life
force, or vitalism, exists in living organisms, this life force cannot be studied by physical or
chemical methods. | d. | living organisms
are composed of the same elements present in nonliving things, plus a few special trace elements
found only in living organisms or their products. | e. | living organisms can be understood in terms of the same physical and chemical
laws that can be used to explain all natural phenomena. |
|
|
|
4.
|
One of the following people set up a closed system
to mimic Earth's early atmosphere and discharged electrical sparks through it. A variety of
organic compounds common in organisms were formed. Who did this?
a. | Stanley Miller | b. | Jakob Berzelius | c. | Friedrich
Wohler | d. | Hermann Kolbe | e. | August Kekulé |
|
|
|
5.
|
Which of the following people used this apparatus
to study formation of organic compounds?
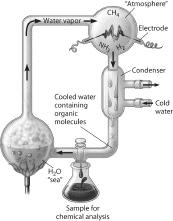
a. | Stanley Miller | b. | Jakob Berzelius | c. | Friedrich
Wohler | d. | Hermann Kolbe | e. | August Kekulé |
|
|
|
6.
|
Which of the following people was the first to
synthesize an organic compound, urea, from inorganic starting materials?
a. | Stanley Miller | b. | Jakob Berzelius | c. | Friedrich
Wohler | d. | Hermann Kolbe | e. | August Kekulé |
|
|
|
7.
|
Which of the following people's synthesis of
this compound from inorganic starting materials provided evidence against vitalism?
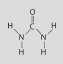
a. | Stanley Miller | b. | Jakob Berzelius | c. | Friedrich
Wohler | d. | Hermann Kolbe | e. | August Kekulé |
|
|
|
8.
|
Which of the following people synthesized an
organic compound, acetic acid, from inorganic substances that had been prepared directly from pure
elements?
a. | Stanley Miller | b. | Jakob Berzelius | c. | Friedrich
Wohler | d. | Hermann Kolbe | e. | August Kekulé |
|
|
|
9.
|
Which of the following people's synthesis of
this compound from inorganic starting materials provided evidence against vitalism?
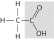
a. | Stanley Miller | b. | Jakob Berzelius | c. | Friedrich
Wohler | d. | Hermann Kolbe | e. | August Kekulé |
|
|
|
10.
|
One of the following people was the first to
suggest that organic compounds, those found in living organisms, were distinctly different from
inorganic compounds found in the nonliving world. Though this suggestion is now known to be
incorrect, it stimulated important research into organic compounds. Who suggested this?
a. | Stanley Miller | b. | Jakob Berzelius | c. | Friedrich
Wohler | d. | Hermann Kolbe | e. | August Kekulé |
|
|
|
11.
|
How many electron pairs does carbon share in order
to complete its valence shell?
|
|
|
12.
|
A carbon atom is most likely to form what kind of
bond(s) with other atoms?
a. | ionic | b. | hydrogen | c. | covalent | d. | A and B
only | e. | A, B, and C |
|
|
|
13.
|
Which of the following statements best describes
the carbon atoms present in a seed-eating bird?
a. | They were incorporated into organic molecules by
plants. | b. | They were processed into sugars through
photosynthesis. | c. | They are
ultimately derived from carbon dioxide. | d. | Only A and C are
correct. | e. | A, B, and C are
correct. |
|
|
|
14.
|
Why are hydrocarbons insoluble in
water?
a. | The majority of their bonds are polar covalent
carbon-to-hydrogen linkages. | b. | The majority of
their bonds are nonpolar covalent carbon-to-hydrogen linkages. | c. | They are hydrophilic. | d. | They exhibit
considerable molecular complexity and diversity. | e. | They are lighter than water. |
|
|
|
15.
|
How many structural isomers are possible for a
substance having the molecular formula 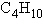 ? ?
|
|
|
16.
|
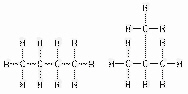
Figure
4.1
The two molecules shown in Figure 4.1 are best described asa. | optical isomers. | b. | radioactive isotopes. | c. | structural
isomers. | d. | nonradioactive isotopes. | e. | geometric isomers. |
|
|
|
Figure
4.2
|
|
|
17.
|
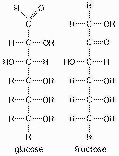
Shown here in Figure 4.2 are the structures of
glucose and fructose. These two molecules differ in the
a. | number of carbon, hydrogen, and oxygen
atoms | b. | types of carbon, hydrogen, and oxygen
atoms. | c. | arrangement of carbon, hydrogen, and oxygen
atoms. | d. | number of oxygen atoms joined to carbon atoms by double
covalent bonds. | e. | answers A, B, and
C |
|
|
|
18.
|
Shown here in Figure 4.2 are the structures of
glucose and fructose. These two molecules are
a. | geometric isotopes. | b. | enantiomers. | c. | geometric
isomers. | d. | structural isomers. | e. | nonisotopic isomers. |
|
|
|
19.
|
Which of the following statements correctly
describes geometric isomers?
a. | They have variations in arrangement around a double
bond. | b. | They have an asymmetric carbon that makes them mirror
images. | c. | They have the same chemical
properties. | d. | They have
different molecular formulas. | e. | Their atoms and
bonds are arranged in different sequences. |
|
|
|
20.
|
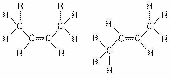
Figure
4.3
The two molecules shown in Figure 4.3 are best described asa. | enantiomers. | b. | radioactive isotopes. | c. | structural
isomers. | d. | nonisotopic isomers. | e. | geometric isomers. |
|
|
|
21.
|
Research indicates that Ibuprofen, a drug used to
relieve inflammation and pain, is a mixture of two enantiomers; that is, molecules that
a. | have identical three-dimensional
shapes. | b. | are mirror images of one
another. | c. | lack an asymmetric carbon. | d. | differ in the location of their double bonds. | e. | differ in their electrical charge. |
|
|
|
22.
|
Research indicates that Albuterol, a drug used to
relax bronchial muscles, improving airflow and thus offering relief from asthma, consists only of one
enantiomer, the R-form. Why is it important for this drug to consist of only one enantiomeric form,
rather than a mixture of enantiomers?
a. | Different enantiomers may have different or opposite
physiological effects. | b. | It is impossible
to synthesize mixtures of enantiomers. | c. | It is much less
expensive to synthesize one enantiomer at a time. | d. | Albuterol is an example of a compound for which only one enantiomer
exists. | e. | Only the R-form of Albuterol has been studied; until
more information is available, physicians prefer to use the pure
R-form. |
|
|
|
23.
|
Three or four of the following illustrations depict
different structural isomers of the organic compound with molecular formula 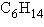 .
For clarity, only the carbon skeletons are shown; hydrogen atoms that would be attached to the
carbons have been omitted. Which one, if any, is NOT a structural isomer of this
compound? .
For clarity, only the carbon skeletons are shown; hydrogen atoms that would be attached to the
carbons have been omitted. Which one, if any, is NOT a structural isomer of this
compound?
a. | 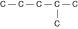 | b. | 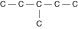 | c. | 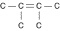 | d. | 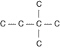 | e. | Each of the illustrations in the other answer choices depicts a structural
isomer of the compound with molecular formula 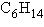 . . |
|
|
|
24.
|
Which of the pairs of molecular structures shown
below depict enantiomers (enantiomeric forms) of the same molecule?
|
|
|
25.
|
Which of the pairs of molecular structures shown
below do NOT depict enantiomers (enantiomeric forms) of the same molecule?
|
|
|
26.
|
Three or four of the pairs of structures shown
below depict enantiomers (enantiomeric forms) of the same molecule. Which pair, if any, are NOT
enantiomers of a single molecule? If each of the pairs depicts enantiomers, choose answer
F.
a. | 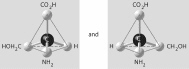 | b. | 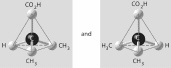 | c. |  | d. |  | e. | 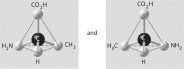 | f. | Both illustrations in each of the other answer choices depict enantiomers of
the same molecule. |
|
|
|
27.
|
Thalidomide and L-dopa, shown below, are examples
of pharmaceutical drugs that occur as enantiomers, or molecules that
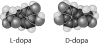
a. | have identical three-dimensional
shapes. | b. | are mirror images of one
another. | c. | lack an asymmetric carbon. | d. | differ in the location of their double bonds. | e. | differ in their electrical charge. |
|
|
|
28.
|
A compound contains hydroxyl groups as its
predominant functional group. Which of the following statements is true concerning this
compound?
a. | It lacks an asymmetric carbon, and it is probably a fat
or lipid. | b. | It should dissolve in water. | c. | It should dissolve in a nonpolar solvent. | d. | It won't form hydrogen bonds with water. | e. | It is hydrophobic. |
|
|
|
29.
|
Which is the best description of a carbonyl
group?
a. | an oxygen joined to a carbon by a single covalent
bond | b. | a nitrogen and two hydrogens joined to a carbon by
covalent bonds | c. | a carbon joined to
two hydrogens by single covalent bonds | d. | a sulfur and a
hydrogen joined to a carbon by covalent bonds | e. | a carbon atom
joined to an oxygen by a double covalent bond |
|
|
|
30.
|
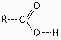
Figure
4.4
What is the name of the functional group shown in Figure 4.4?a. | carbonyl | b. | ketone | c. | aldehyde | d. | carboxyl | e. | hydroxyl |
|
|
|
31.
|
Which of the following contains nitrogen in
addition to carbon, oxygen, and hydrogen?
a. | an alcohol such as ethanol | b. | a monosaccharide such as glucose | c. | a steroid such as testosterone | d. | an amino acid such as glycine | e. | a hydrocarbon such as benzene |
|
|
|
32.
|
Which of the following is a false statement
concerning amino groups?
a. | They are basic in pH. | b. | They are found in amino acids. | c. | They contain nitrogen. | d. | They are
nonpolar. | e. | They are components of
urea. |
|
|
|
33.
|
Which two functional groups are always found
in amino acids?
a. | ketone and aldehyde | b. | carbonyl and carboxyl | c. | carboxyl and
amino | d. | phosphate and sulfhydryl | e. | hydroxyl and aldehyde |
|
|
|
34.
|
Amino acids are acids because they always possess
which functional group?
a. | amino | b. | carbonyl | c. | carboxyl | d. | sulfhydryl | e. | aldehyde |
|
|
|
35.
|
A carbon skeleton is covalently bonded to both an
amino group and a carboxyl group. When placed in water it
a. | would function only as an acid because of the carboxyl
group. | b. | would function only as a base because of the amino
group. | c. | would function as neither an acid nor a
base. | d. | would function as both an acid and a
base. | e. | is impossible to determine how it would
function. |
|
|
|
36.
|
A chemist wishes to make an organic molecule less
acidic. Which of the following functional groups should be added to the molecule in order to do
so?
a. | carboxyl | b. | sulfhydryl | c. | hydroxyl | d. | amino | e. | phosphate |
|
|
|
37.
|
Which functional groups can act as
acids?
a. | amine and sulfhydryl | b. | carbonyl and carboxyl | c. | carboxyl and
phosphate | d. | hydroxyl and aldehyde | e. | ketone and amino |
|
|
|
The following questions refer to the structures
shown in Figure 4.5.
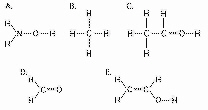
Figure 4.5
|
|
|
38.
|
Which of the structures is an impossible covalently
bonded molecule?
|
|
|
39.
|
Which of the structures contain(s) a carboxyl
functional group?
a. | A | b. | B | c. | C | d. | C and
E | e. | none of the
structures |
|
|
|
40.
|
In which of the structures are the atoms bonded by
ionic bonds?
a. | A | b. | B | c. | C | d. | C, D, and E
only | e. | none of the
structures |
|
|
|
41.
|
Testosterone and estradiol are
a. | nucleic acids. | b. | carbohydrates. | c. | proteins. | d. | phospholipids. | e. | steroids. |
|
|
|
42.
|
Testosterone and estradiol are male and female sex
hormones, respectively, in many vertebrates. In what way(s) do these molecules differ from each
other?
a. | Testosterone and estradiol are structural isomers but
have the same molecular formula. | b. | Testosterone and
estradiol are geometric isomers but have the same molecular formula. | c. | Testosterone and estradiol have different functional groups attached to the
same carbon skeleton. | d. | Testosterone and
estradiol have distinctly different chemical structures, with one including four fused rings of
carbon atoms, while the other has three rings. | e. | Testosterone and
estradiol are enantiomers of the same organic molecule. |
|
|
|
43.
|
Organic chemistry is currently defined
as
a. | the study of compounds made only by living
cells. | b. | the study of carbon
compounds. | c. | the study of vital
forces. | d. | the study of natural (as opposed to synthetic)
compounds. | e. | the study of
hydrocarbons. |
|
|
|
44.
|
Which of the following hydrocarbons has a double
bond in its carbon skeleton?
|
Short Answer
|
|
|
The following questions refer to the functional
groups shown in Figure 4.6.
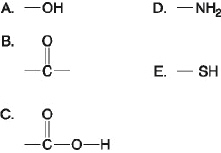
Figure
4.6
|
|
|
45.
|
Which is a hydroxyl functional group?
|
|
|
46.
|
Which is an amino functional group?
|
|
|
47.
|
Which is a carbonyl functional group?
|
|
|
48.
|
Which is a functional group that helps stabilize
proteins by forming covalent cross-links within or between protein molecules?
|
|
|
49.
|
Which is a carboxyl functional group?
|
|
|
50.
|
Which is an acidic functional group that can
dissociate and release H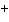 into a solution? into a solution?
|
|
|
51.
|
Which is a basic functional group that can accept
H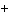 and become positively charged? and become positively charged?
|
|
|
The following questions refer to the molecules
shown in Figure 4.7.
Figure 4.7
|
|
|
52.
|
Which molecule is water soluble because it has a
hydroxyl functional group?
|
|
|
53.
|
Which molecule is an alcohol?
|
|
|
54.
|
Which molecules contain a carbonyl group?
A) A
and B
B) B and C
C) C and D
D) D and E
E) E and A
|
|
|
55.
|
Which molecule has a carbonyl functional group in
the form of a ketone?
|
|
|
56.
|
Which molecule has a carbonyl functional group in
the form of an aldehyde?
|
|
|
57.
|
Which molecule contains a carboxyl
group?
|
|
|
58.
|
Which molecule can increase the concentration of
hydrogen ions in a solution and is therefore an organic acid?
|
|
|
The following questions refer to the molecules
shown in Figure 4.8.
Figure 4.8
|
|
|
59.
|
Which molecule contains a sulfhydryl functional
group?
|
|
|
60.
|
Which molecule functions to transfer energy between
organic molecules?
|
|
|
61.
|
Which molecule contains an amino functional group,
but is not an amino acid?
|
|
|
62.
|
Which molecule is a thiol?
|
|
|
63.
|
Which molecule is an organic
phosphate?
|
|
|
64.
|
Which molecule can function as a
base?
|
|
|
65.
|
Identify the asymmetric carbon in this
molecule:
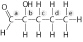
|
Multiple Response
Identify one
or more choices that best complete the statement or answer the question.
|
|
|
66.
|
Choose the term that correctly describes the
relationship between these two sugar molecules:
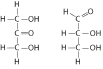
|
|
|
67.
|
Which functional group is not present in
this molecule?
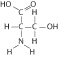
|
|
|
68.
|
Which action could produce a carbonyl
group?
|
|
|
69.
|
Which chemical group is most likely to be
responsible for an organic molecule behaving as a base?
|